Contributing author: Lars Johansson, CSO @ Antaros Medical
Chronic liver disease, and its advanced condition of cirrhosis, are both a growing worldwide healthcare problem and a highly active field of research.
Global mortality due to liver cirrhosis and chronic liver diseases is rising, though there has been a shift over time in the aetiologies that are driving these numbers. While we have gotten better at treating hepatitis B and C with antivirals, increasing prevalence of metabolic dysfunction-associated liver disease (MASLD) / metabolic dysfunction-associated steatohepatitis (MASH), and MetALD, where they co-exist with increased alcohol consumption, are rising in prevalence.
This blog post will cover the complexity of chronic liver disease and cirrhosis – its drivers, progression via inflammation and fibrosis, and the implications for drug development and treatment approaches. It will also cover some of the biomarkers and non-invasive tests (NITs) currently available and in development that can help to further our disease understanding, select and enrich our patient populations, and ultimately provide insights to help advance drug development in this space.
Quentin Anstee, Pro-Vice-Chancellor of the Faculty of Medical Sciences at Newcastle University, and I discussed this and more in a webinar called ‘Cracking Cirrhosis: Complexity of chronic liver disease and insights for drug development’.
Different aetiologies but a common pathophysiological pathway to cirrhosis
Liver cirrhosis is the result of different causes of chronic liver injury, including metabolic dysfunction (as in MASLD/MASH), viral infection (e.g. hepatitis B and C), autoimmune disorders, genetic conditions, drug-related injury, or lifestyle drivers (as in alcohol-related liver disease; ALD). These different drivers and aetiologies of chronic liver disease result in similar patterns of progression via a common pathophysiological pathway.
In this pathway, a healthy liver is injured in some way (e.g. steatosis, viral infection, etc.), which initiates an inflammatory process which, if sustained over a longer period, will result in the formation of scar tissue. Once enough scar tissue has been deposited and cirrhosis is established, if the number of functioning hepatocytes continues to decrease this can lead to decompensation or hepatocellular carcinoma (HCC).
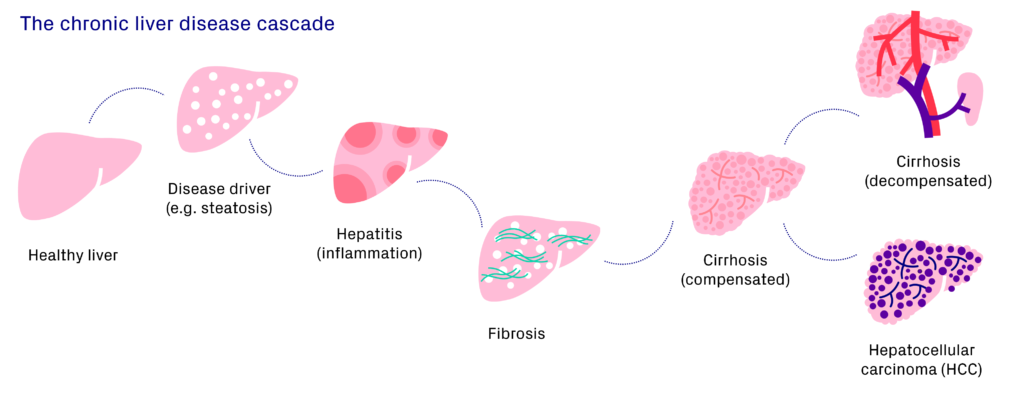
Initiation of inflammation and fibrosis pathways
Progression to cirrhosis is via inflammation and fibrosis. The pathways involved in inflammation and the formation of fibrosis are incredibly complex, a simplified version is described below:
- The hepatocyte injury (which in addition to the aetiologies already mentioned can also be caused by extrahepatic signals from the adipose tissue, intestine, bile duct or muscle) causes hepatic stellate cells (HSCs) to activate and differentiate into myofibroblasts.
- This can also happen via the inflammatory pathway. Activation of macrophages leads to the recruitment of immune cells. Signals from the macrophages and the recruited immune cells (such as neutrophils and monocytes) stimulate the activation of HSCs.
- The HSCs also have an autocrine signalling loop, signalling to themselves to activate and differentiate into myofibroblasts.
- The activated myofibroblasts then produce and deposit extracellular matrix (ECM) within the liver.
- The ECM accumulates and replaces the normal parenchyma with connective tissue
The good news is that liver fibrosis is reversible. In short, upon removal of the hepatocyte injury there is reduced cytokine signalling and apoptosis or de-differentiation of myofibroblasts. ECM production is slowed, the collagen fibres are degraded and, at least in early stages of fibrosis, this can lead to fibrosis resolution. This has been shown, for example, in both bariatric surgery and with antiviral treatment for hepatitis C virus (HCV), where there can be significant effects if the cause of the injury is removed.
However, at a certain stage of fibrosis, even if the cause of the injury is removed, the fibrosis still progresses. One hypothesis to address this is the idea that there are hot and cold forms of fibrosis. ‘Hot’ fibrosis describes an inflammatory state with lots of immune cell activity, while ‘cold’ fibrosis refers to fibrosis with less inflammatory activity. This will be a very important concept for drug development, and an exciting avenue for future research.
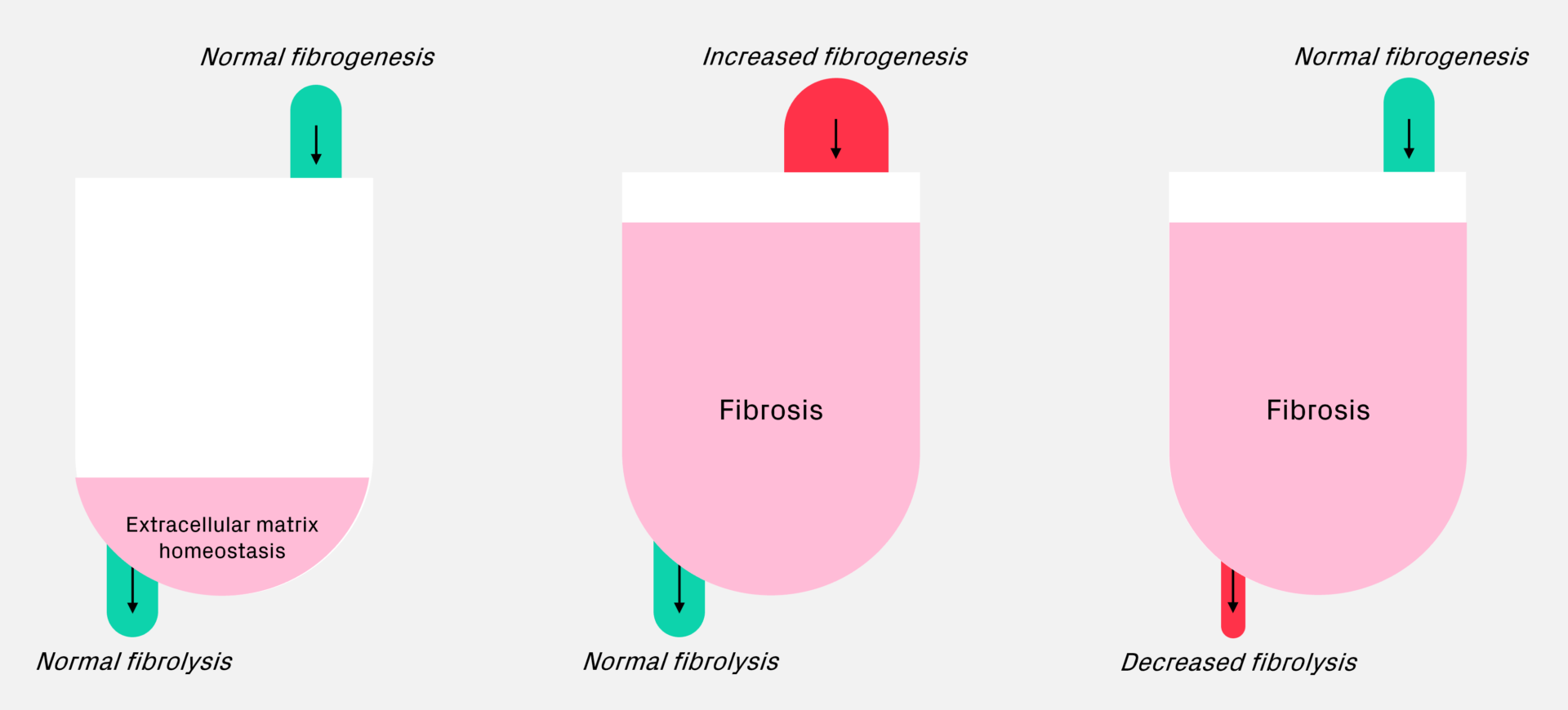
Fibrosis: a balance between fibrogenesis and fibrolysis
Under normal conditions, fibrogenesis (the formation of fibrosis) and fibrolysis (resolution of fibrosis) are balanced in extracellular matrix homeostasis. If there is increased fibrogenesis, you see the metaphorical bathtub start to fill up, representing a build-up of fibrosis in the liver. And vice versa if you have normal fibrogenesis with reduced fibrolysis, this can also lead to build-up of fibrosis. This model can be very useful for describing where different interventions are acting in the process and what treatment response you could expect to see.
Biomarkers for assessing inflammation, fibrosis, and cirrhosis in drug development
Historically, histology has been considered the ‘gold standard’ for assessing inflammation and fibrosis in drug development. It has several important limitations such as sampling error, observer variability, and in addition to being resource-intensive and costly, it is invasive and carries risk of complications.
This has created a need for non-invasive tests (NITs), alternatives to biopsy that can be used for diagnosis, monitoring, and quantifying treatment response. These NITs can be sorted broadly into circulating panels, liver stiffness measurements and imaging modalities (such as magnetic resonance imaging; MRI, and positron emission tomography; PET), each with its advantages and disadvantages.
Cirrhosis drug development and treatment approaches
As the fibrosis in the liver accumulates and scar tissue increasingly replaces normal tissue, cirrhosis can develop. In compensated cirrhosis, the liver can maintain its important functions and any observed symptoms are generally non-specific. In decompensated cirrhosis, however, the liver no longer functions adequately, portal hypertension develops, and this can lead to several complications such as ascites, variceal bleeding, and hepatic encephalopathy.
Treatment approaches for cirrhosis
There are different ways to approach treating of cirrhosis based on targeting different stages along the disease progression pathway. The first step is addressing the underlying aetiology and removing the cause of the hepatocyte injury (e.g. antiviral therapy, abstinence from alcohol, or inducing weight loss). The next steps are slowing progression and promoting regression, either sequentially or, perhaps in the future, simultaneously. This is a very exciting area of research currently, and the target of many potential treatments in development. The last step in cases of advanced disease is preventing decompensation events and providing symptomatic relief for complications.
Major challenges in drug development for chronic liver disease
There are many challenges facing chronic liver disease and cirrhosis drug development. Some of the major ones include a heterogeneous patient population, high placebo response rate, sampling error of biopsy and other biomarkers, and the overall complexity of the disease.
Chronic liver disease complexity – bringing it all together
One way to think about how this all fits together is to consider the different disease drivers at across the various stages of disease progression. This can help provide a framework for thinking about what we are trying to treat and when are we trying to treat it, and what we are trying to measure and when are we trying to measure it.
Using MASLD/MASH as an example, you could look at steatosis, inflammation, fibrogenesis, portal pressure and hepatocyte function in relation to disease state. The curves are not exact mathematics, but rather represent the concept of how the involvement of these different processes at different stages of disease will influence both the treatment goals and the suitable response biomarkers.
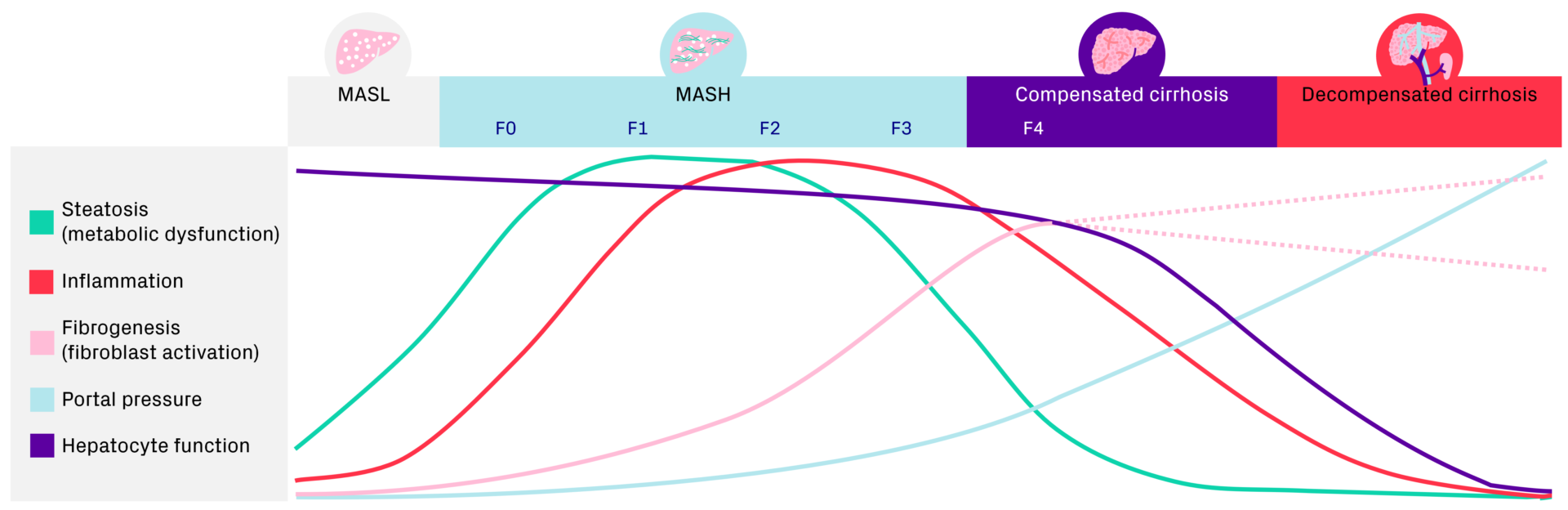
This example can also help put into context how we think about new and novel biomarkers. For example, gadoxetate MRI offers a potential solution to non-invasively assess changes in liver function, which could provide more valuable insights from fibrosis grade F2 and onwards, where you can expect there will be changes in hepatocyte function. Or fibrogenesis PET imaging with PDGFRß, which can be used to detect changes in fibrogenesis activity much earlier and faster than it would take to detect meaningful changes in fibrosis using other methods. This information may be more useful during earlier stages of the disease, as fibrogenesis activity is increasing.
One simple biomarker that could be useful throughout all disease stages and in evaluating disease severity is the liver/spleen volume ratio. As chronic liver disease progresses, the right lobe of the liver decreases while the left lobe increases, and the spleen also increases. The ratio of the volumes can help us to understand what is happening and if there are any confounding factors.
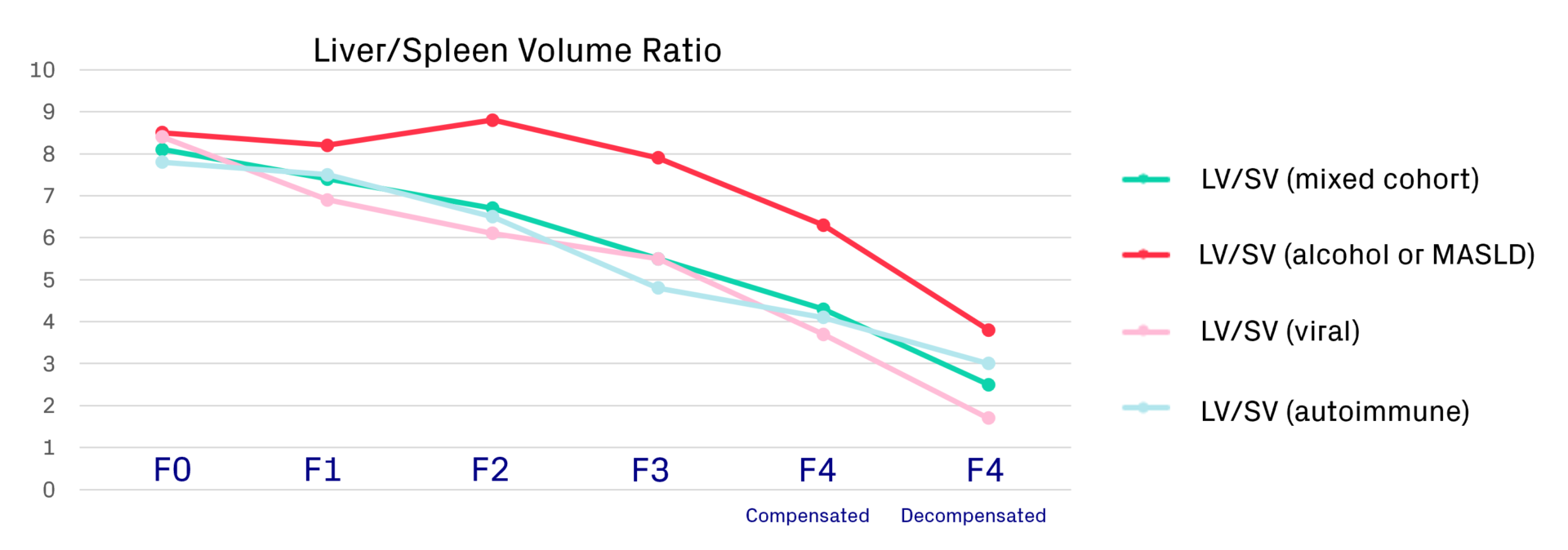
We recently presented an abstract at the AASLD meeting on how to correct for steatosis in the liver/spleen volume ratio for MASLD to normalise that curve so that it would follow the same pattern as with the other aetiologies.
In Summary
To summarise briefly what has been covered in this blog post:
- Chronic liver disease is complex and there is still more to be learnt about the pathophysiological processes driving disease progression
- Biomarkers (circulating and imaging) can help us to better understand disease progression/regression and provide valuable insights into treatment responses
- The complexity of the disease makes drug development in this space challenging, but it is achievable. Thoughtful patient selection, deep understanding of the disease, and validation of non-invasive biomarkers are crucial for meeting these challenges
Blog disclaimer
The views and opinions expressed in this article are solely those of the contributing author/s. These views and opinions do not necessarily represent those of Antaros Medical.
Contact details
If you have any questions regarding this article, please reach out to press@antarosmedical.com
